What is the foundation of a successful Phase II clinical trial?
In drug development, Phase II is the critical bridge that validates concepts and assesses efficacy. Its success is never accidental; it is deeply rooted in a rigorous data chain from preclinical stages through Phase I, and fundamentally dependent on high-purity, highly consistent research materials — an area where partners like PHMO specialize in supporting translational and early clinical research.
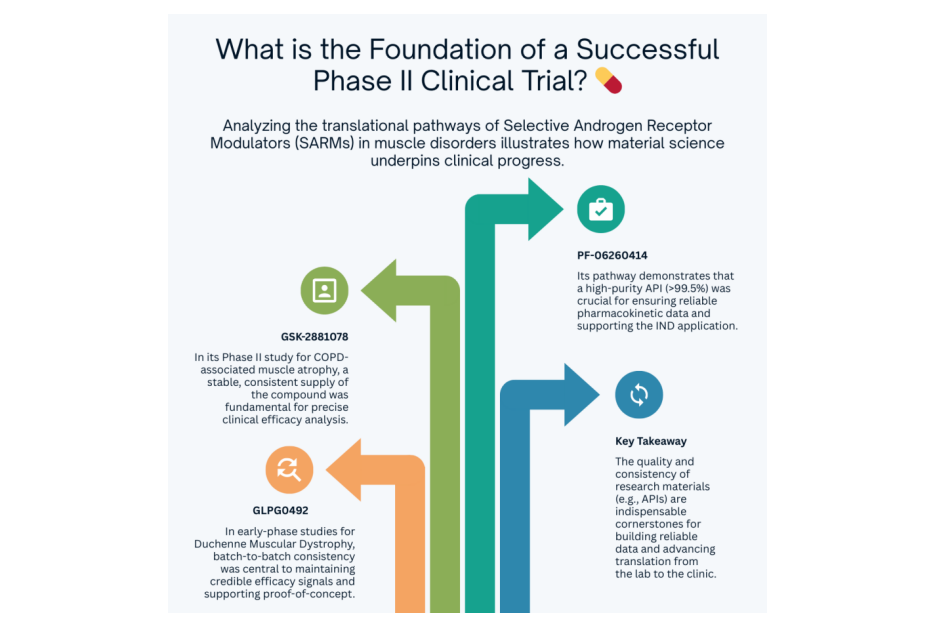
Taking the cutting-edge Selective Androgen Receptor Modulators (SARMs) in muscle disorders as an example, analyzing the translational pathways of PF-06260414, GSK-2881078, and GLPG0492 clearly shows how material science escorts clinical progress:
➠PF-06260414 (Pfizer): Its preclinical data demonstrated exceptional muscle selectivity. Building on this, a Phase I trial (NCT02175134) showed good tolerability at doses of 1-100 mg/day, paving the way to explore its potential in cancer cachexia, particularly for chemotherapy-induced muscle wasting. Throughout this process, high-purity API (>99.5%) served as the invisible pillar ensuring reliable pharmacokinetic data, supporting the IND application, and advancing toward potential Phase II studies.
➠GSK-2881078 (GSK): It advanced directly into a Phase II study (NCT03359473) for COPD-associated muscle atrophy and cancer cachexia. The ingenuity of its translational logic lies not only in aiming to improve muscle function but also in integrating potential anti-inflammatory mechanisms to address concerns about cardiovascular side effects in chronic disease treatment. A stable, consistent supply of the compound (within the 0.5-2 mg/day dose range) is fundamental for precisely parsing its clinical efficacy and distinguishing it from purely anabolic SARMs.
➠ GLPG0492 (Galapagos): The Phase I/II study (NCT02124005) for Duchenne Muscular Dystrophy (DMD) addresses the urgent need for non-injectable therapies in genetic muscle disorders. Preclinical models showed its ability to enhance strength and reduce fat accumulation. From lab to early-stage clinic, batch-to-batch consistency is the lifeline maintaining the credibility of its efficacy signal and supporting the proof-of-concept for this regenerative approach.
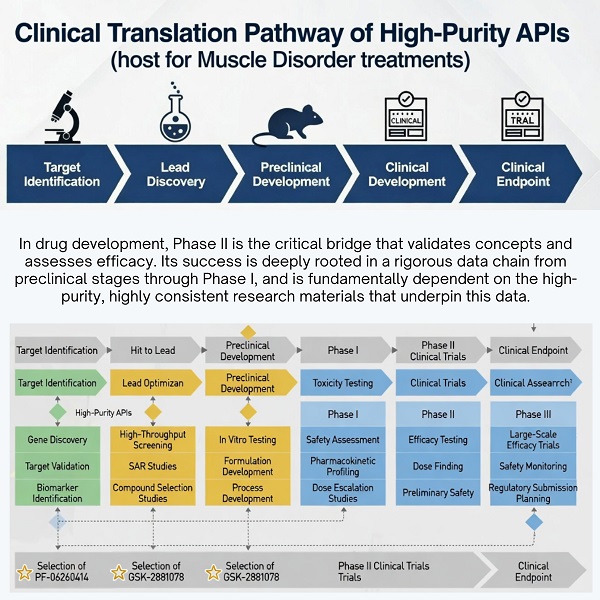
A common thread in these cases is the critical role of high-purity (>99.5%), batch-consistent APIs—such as those supplied by partners like PHMO.
At PHMO, we work with global biotech and pharma teams to ensure this foundational consistency from research supply through clinical translation.
Your perspective: What is the most significant non-technical challenge in moving a novel target from lab to clinic? Navigating regulatory pathways, securing sustained funding, or foundational elements like API quality/consistency?
References:
1.https://clinicaltrials.gov/study/NCT02175134
2.https://clinicaltrials.gov/study/NCT03359473
3.https://clinicaltrials.gov/study/NCT02124005
