
Brensocatib
A small molecule inhibitor of dipeptidyl peptidase 1 (DPP1), an enzyme inhibitor. It is used to reduce the risk of exacerbations in patients with non-cystic fibrosis bronchiectasis (NCFB) by inhibiting neutrophil-mediated inflammation.
Overview
Brensocatib is a first-in-class oral inhibitor of dipeptidyl peptidase 1 (DPP1) that modulates inflammatory responses by inhibiting neutrophil serine protease activation. It reduces neutrophilic inflammatory burden in patients with non-cystic fibrosis bronchiectasis (NCFB), offering a targeted approach to manage disease progression.
Synonyms: CS-2785; AZD7986; INS1007; INS-1007; Brensocatib
Product Categories: Azepines; BCRP/ABCG2 Inhibitors; BCRP/ABCG2 Substrates; Cytochrome P-450 Enzyme Inducers; Heterocyclic Compounds, Fused-Ring; MATE 1 Inhibitors; OATP1B1/SLCO1B1 Inhibitors; P-glycoprotein substrates
Mol File: 1802148-05-5.mol
Physicochemical Properties
Storage temp: -20°C
Solubility: DMSO: 100.0(Max Conc. mg/mL);237.83(Max Conc. mM)
Form: Solid
Color: Light Yellow to Yellow
Water Solubility: Slightly soluble at pH 1.2; freely soluble at pH 4.5; very slightly soluble at pH 6.8
MSDS Information
Experimental Data
1. Cell Experiment
Please select an appropriate solvent for preparing stock solutions based on the product's solubility in different solvents. Once prepared, the solution should be aliquoted and stored to avoid product degradation caused by repeated freeze-thaw cycles. Storage conditions and duration for stock solutions: -80°C, 2 years; -20°C, 1 year. When stored at -80°C, use within 2 years; when stored at -20°C, use within 1 year.
Preparing Stock Solutions:

2. Animal Experiment
Please select an appropriate dissolution protocol based on your experimental animals and route of administration. For the following protocols, first prepare a clear stock solution following the in vitro method, then add cosolvents sequentially:
To ensure experimental reliability, the clear stock solution can be appropriately stored according to storage conditions. For in vivo working solutions, it is recommended to prepare them fresh and use on the same day. The percentage indicated before each solvent represents its volume ratio in the final solution. If precipitation occurs during preparation, heating and/or ultrasonication can be used to aid dissolution.
Protocol 1
Add solvents in order: 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% saline
Solubility: ≥ 2.5 mg/mL (5.95 mM); Clear solution
This protocol yields a clear solution of ≥ 2.5 mg/mL (5.95 mM, saturation unknown).
Example for 1 mL working solution: Add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix well. Add 50 μL Tween-80 to the mixture and mix thoroughly. Then add 450 μL saline to bring the volume to 1 mL.
Protocol 2
Add solvents in order: 10% DMSO → 90% corn oil
Solubility: ≥ 2.5 mg/mL (5.95 mM); Clear solution
This protocol yields a clear solution of ≥ 2.5 mg/mL (5.95 mM, saturation unknown). This method is not suitable for experiments lasting longer than half a month.
Example for 1 mL working solution: Add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL corn oil and mix well.
Pharmacodynamics
Brensocatib is a competitive, reversible inhibitor of DPP1, leading to decreased NSP activity in patients. Exploratory assays showed brensocatib was associated with dose-dependent reductions in NSP activity in patients with NCFB. In a phase 1 study, brensocatib was associated with dose-dependent reductions in neutrophil elastase activity in the blood. A population pharmacokinetic/pharmacodynamic analysis revealed a threshold brensocatib exposure (AUC) effect for attaining sputum neutrophil elastase (NE) levels below the limit of quantification, and a strong relationship between sputum NE levels below the limit of quantification and a reduction in pulmonary exacerbations.
Treatment with brensocatib is associated with dermatologic adverse reactions and gingival/periodontal adverse reactions. The use of live attenuated vaccines should be avoided.
Mechanism Of Action
Non-cystic fibrosis bronchiectasis (NCFB) is a chronic lung disease characterized by a cycle of infection, inflammation, and lung tissue damage. Pharmacologic treatment of NCFB is targeted against dipeptidyl peptidase 1 (DPP1), a cysteine protease which plays a crucial role in the activation of pro-inflammatory neutrophil serine proteases (NSPs) during neutrophil maturation in the bone marrow - including cathepsin G (CatG), neutrophil elastase (NE) and proteinase 3 (PR3) - which are implicated in the pathogenesis of neutrophil-mediated NCFB inflammation. Brensocatib is a competitive, reversible inhibitor of dipeptidyl peptidase 1 (DPP1). In cell-based assays, DPP1 inhibition by brensocatib reduces the activity of NSPs including neutrophil elastase, cathepsin G, and proteinase 3.
Similar Products
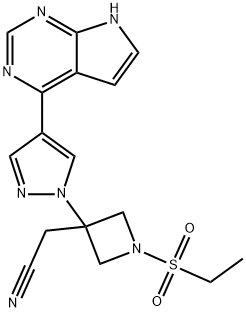
Baricitinib
Therapy
: Immune-Inflammatory diseases
Target
: JAK1;JAK2
CAS
: 1187594-09-7
MF
: C16H17N7O2S
MW
: 371.42
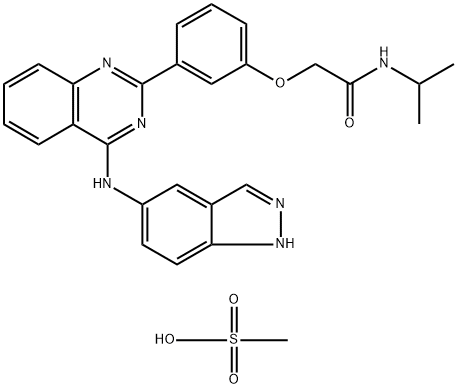
Belumosudil Mesylate
Therapy
: Immune-Inflammatory diseases
Target
: ROCK
CAS
: 2109704-99-4
MF
: C27H28N6O5S
MW
: 548.62
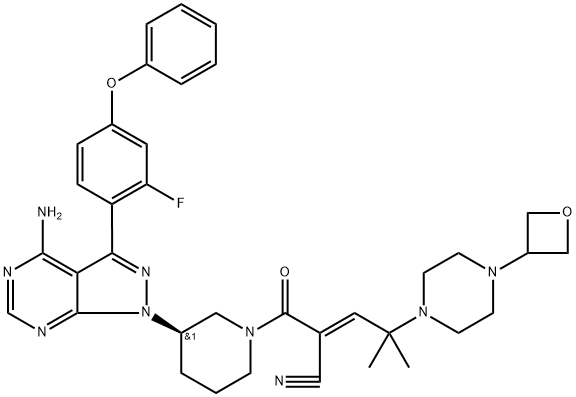
Rilzabrutinib
Therapy
: Immune-Inflammatory diseases
Target
: BTK
CAS
: 1575596-29-0
MF
: C36H40FN9O3
MW
: 665.77
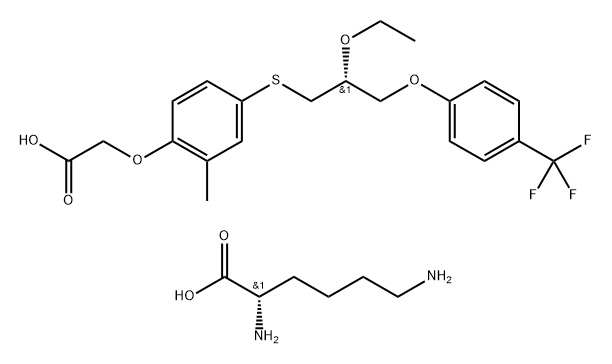
Seladelpar lysine
Therapy
: Immune-Inflammatory diseases
Target
: PPARδ
CAS
: 928821-40-3
MF
: C27H37F3N2O7S
MW
: 590.66
