
AFICAMTEN
A selective allosteric inhibitor of cardiac myosin; a cardiovascular therapeutic agent, used for the treatment of symptomatic obstructive hypertrophic cardiomyopathy (oHCM).
Overview
AFICAMTEN is a potent oral non-steroidal cardiac myosin inhibitor, which has the effects of inhibiting cardiac myosin ATPase activity and regulating myocardial contractility. It can be used for the research on hypertrophic cardiomyopathy, heart failure and other related cardiovascular diseases.
Synonyms: CK-3773274; BAY-3723113; CK-274
Product Categories: Heterocyclic Compounds; Aromatics; Pharmaceuticals; Intermediates & Fine Chemicals; API
Mol File: 2364554-48-1.mol
Physicochemical Properties
Storage temp: -20℃
Solubility: DMSO: 125 mg/mL (370.50 mM; Need ultrasonic)
Form: Solid
Color: White to Light Brown
MSDS Information
Experimental Data
1. Cell Experiment
DMSO : 125 mg/mL (370.50 mM; Need ultrasonic; Hygroscopic DMSO has a significant impact on the solubility of product, please use newly opened DMSO)
Preparing Stock Solutions:
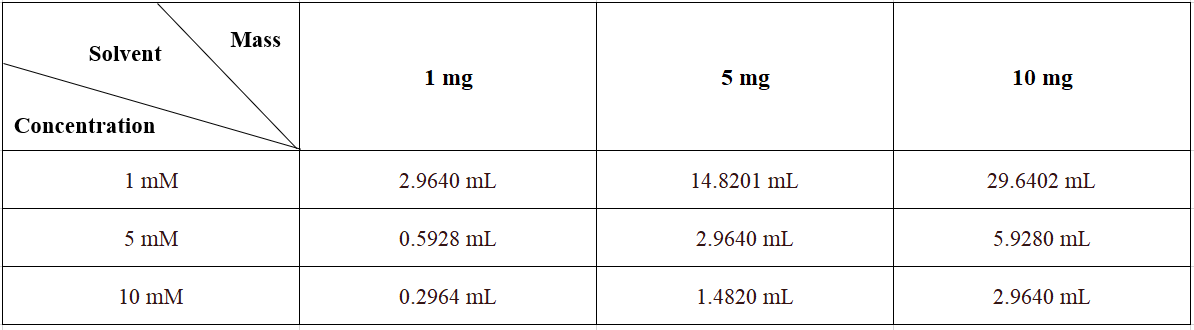
Please refer to the solubility information to select the appropriate solvent. Once prepared, please aliquot and store the solution to prevent product inactivation from repeated freeze-thaw cycles.
Storage method and period of stock solution: -80°C, 6 months; -20°C, 1 month. When stored at -80°C, please use it within 6 months. When stored at -20°C, please use it within 1 month.
2. Animal Experiment
Select the appropriate dissolution method based on your experimental animal and administration route.
For the following dissolution methods, please ensure to first prepare a clear stock solution using an In Vitro approach and then sequentially add co-solvents:
To ensure reliable experimental results, the clarified stock solution can be appropriately stored based on storage conditions. As for the working solution for in vivo experiments, it is recommended to prepare freshly and use it on the same day.
The percentages shown for the solvents indicate their volumetric ratio in the final prepared solution. If precipitation or phase separation occurs during preparation, heat and/or sonication can be used to aid dissolution.
Protocol 1
Add each solvent one by one: 10% DMSO, 40% PEG300, 5% Tween-80, 45% Saline
Solubility: ≥ 2.08 mg/mL (6.17 mM); Clear solution
This protocol yields a clear solution of ≥ 2.08 mg/mL (saturation unknown).
Taking 1 mL working solution as an example, add 100 μL DMSO stock solution (20.8 mg/mL) to 400 μL PEG300, and mix evenly; then add 50 μL Tween-80 and mix evenly; then add 450 μL Saline to adjust the volume to 1 mL.
Preparation of Saline: Dissolve 0.9 g sodium chloride in ddH₂O and dilute to 100 mL to obtain a clear Saline solution.
Protocol 2
Add each solvent one by one: 10% DMSO, 90% (20% SBE-β-CD in Saline)
Solubility: ≥ 2.08 mg/mL (6.17 mM); Clear solution
This protocol yields a clear solution of ≥ 2.08 mg/mL (saturation unknown).
Taking 1 mL working solution as an example, add 100 μL DMSO stock solution (20.8 mg/mL) to 900 μL 20% SBE-β-CD in Saline, and mix evenly.
Preparation of 20% SBE-β-CD in Saline (4°C, storage for one week): 2 g SBE-β-CD powder is dissolved in 10 mL Saline, completely dissolve until clear.
Protocol 3
Add each solvent one by one: 10% DMSO, 90% Corn Oil
Solubility: ≥ 2.08 mg/mL (6.17 mM); Clear solution
This protocol yields a clear solution of ≥ 2.08 mg/mL (saturation unknown). If the continuous dosing period exceeds half a month, please choose this protocol carefully.
Taking 1 mL working solution as an example, add 100 μL DMSO stock solution (20.8 mg/mL) to 900 μL Corn oil, and mix evenly.
Pharmacodynamics
Mechanism Of Action
Similar Products
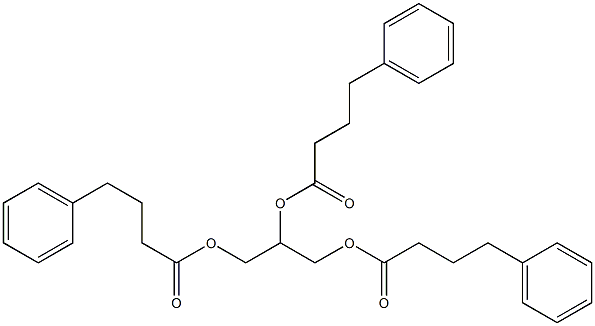
Glycerol Phenylbutyrate
Therapy
: Orphan diseases
Target
: Sigma Receptor
CAS
: 611168-24-2
MF
: C33H38O6
MW
: 530.65
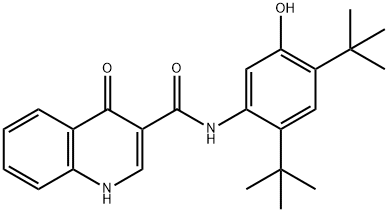
Ivacaftor
Therapy
: Orphan diseases
Target
: CFTR
CAS
: 873054-44-5
MF
: C24H28N2O3
MW
: 392.49
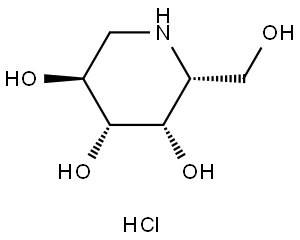
Migalastat Hydrochloride
Therapy
: Orphan diseases
Target
: α-Gal A
CAS
: 75172-81-5
MF
: C6H13NO4.ClH
MW
: 199.63

Tezacaftor
Therapy
: Orphan diseases
Target
: CFTR
CAS
: 1152311-62-0
MF
: C26H27F3N2O6
MW
: 520.5
