All
10 articles in total

JACS: A Novel MSH-Based Skeletal Editing Strategy for Pyridine-to-Pyridazine Transformation – Essential Synthetic Insights!
A carbon-to-nitrogen exchange-based skeletal editing method for converting pyridines to pyridazines has been successfully established. This approach employs MSH as the nitrogen source and utilizes mCPBA to promote ring remodeling. It operates under mild conditions, requires no inert atmosphere or light irradiation, and exhibits excellent functional group tolerance. Furthermore, the method has been applied to the late-stage modification of complex drug molecules, serving as a powerful tool for diversifying heterocycle chemistry and advancing drug discovery.
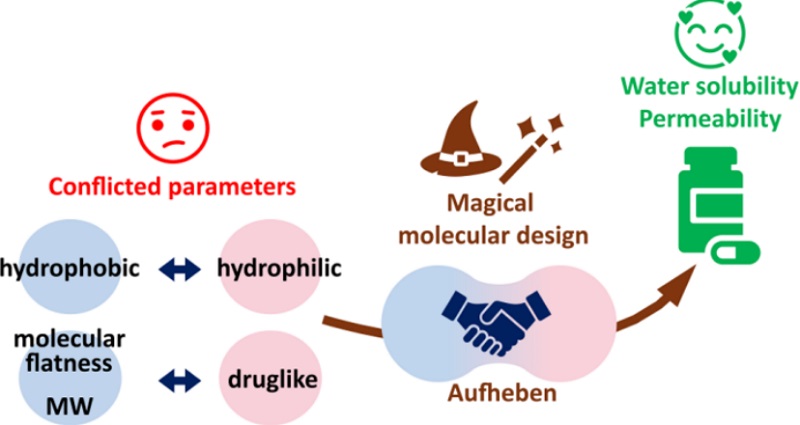
Aufheben Strategy: Resolving Conflicting Properties in Drug Design
The Aufheben strategy enables balanced drug properties by concurrently preserving active conformations and modifying conflicting characteristics. This approach systematically optimizes both small molecules (via disrupted intermolecular interactions) and bRo5 compounds (including cyclic peptides and PROTACs through conformational regulation), offering transformative solutions beyond conventional druglikeness paradigms and expanding molecular design possibilities.
ADC Cytotoxins: The Selection Logic of Precision Anti-Cancer "Warheads"
Antibody-drug conjugates (ADCs), often referred to as "biological missiles" in precision cancer treatment, rely heavily on their "warheads"—small molecule cytotoxins—to determine their tumor-killing efficacy. This article systematically analyzes the core selection criteria for ADC cytotoxins, details the mechanisms of action, structural characteristics, and clinical applications of microtubule inhibitors and DNA damaging agents (two major types), provides a comprehensive overview of the ADC pipeline's cytotoxin coverage, compares the IC50 (half-maximum inhibitory concentration) activity differences of common toxins, and highlights the current challenges of limited toxin variety and singular mechanisms of action, offering crucial insights for ADC drug development.
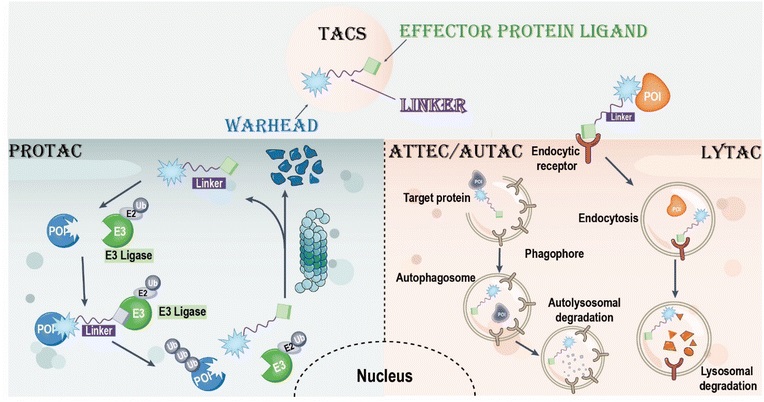
Review of the Development History of TACs Multifunctional Linkers
Targeted chimeras (TACs, such as PROTACs and LYTACs) represent a potential strategy for the selective degradation of difficult-to-drug proteins, acting through bifunctional molecules or molecular glues to provide novel therapies for diseases that are difficult to treat with traditional small molecules. As a bridge connecting target protein ligands and effector protein ligands, the linker of TACs is crucial to molecular conformation and activity. Recent advancements in photo-controlled and cleavable linker design have improved the efficiency and spatiotemporal control of TACs, but challenges remain, such as optimizing stability.
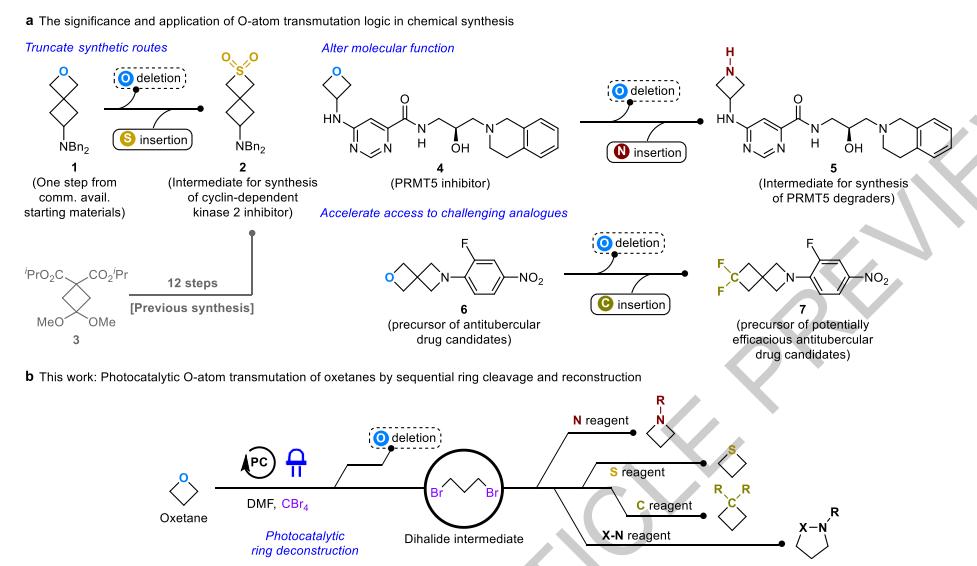
Nature: Photocatalysis Replaces 8-12 Steps with One in Oxetane Editing
Photocatalytic oxetane editing enables selective O-atom replacement with N, S, or C groups in one step. This versatile method simplifies drug synthesis, supports late-stage functionalization, and exhibits broad functional group tolerance, streamlining access to valuable saturated cyclic scaffolds.

Palladium-Catalyzed C-S Coupling Breakthrough Enables Efficient Synthesis of JAK Inhibitor GDC-9918
A scientific research team has made a major breakthrough in the production process of the JAK1/2 inhibitor, GDC-9918. They have successfully resolved two key challenges in large-scale manufacturing, paving the way for the drug's industrialization.
