All
20 articles in total
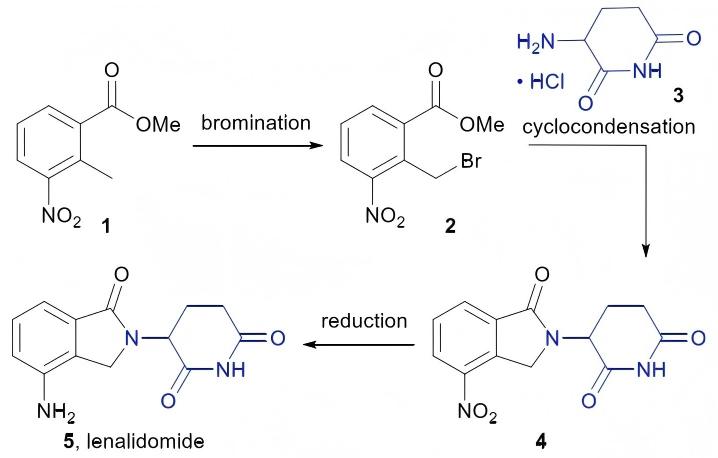
Sustainable Innovation in Pharma: Green Synthesis Process for Lenalidomide API
A major breakthrough has recently been achieved in the research of green synthesis process improvement for lenalidomide active pharmaceutical ingredient. This process perfectly integrates environmental friendliness and production efficiency, effectively solving the environmental problems and cost pain points in traditional production routes, and providing an important practical example for the green transformation of the pharmaceutical industry. The relevant research results have been published in "Org. Process Res. Dev."

Six Major Innovations in Organic Synthesis in October, Enabling New Breakthroughs in Drug Development
Here are some innovative synthetic chemistry technologies to watch in October, recommended by CRO Charles River.
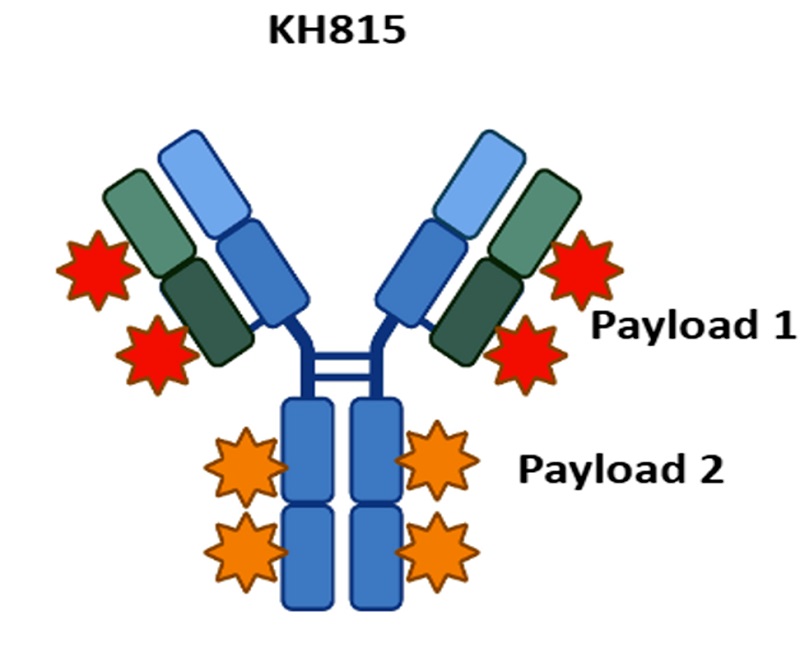
The World's First Dual-load ADC hRS7-E+T (KH815) Preclinical Data Released
Kanghong Pharmaceutical's jointly developed world's first dual-load TROP2-targeted ADC, hRS7-E+T (KH815), published its preclinical results in 《Molecular Cancer Therapeutics》. Its innovative "one antibody, two toxicities" design releases two payloads sequentially, first overcoming drug resistance and then precisely killing the target. In vitro and in vivo experiments (including drug-resistant models) showed superior activity compared to existing ADCs, with an HNSTD of 50 mg/kg. The drug has received clinical trial approvals in China and Australia and is currently initiating international multi-center clinical trials, providing a new solution for tumor drug resistance.
Sevabertinib: A Reversible EGFR-HER2 Inhibitor as a Game-Changer for HER2-Mutant Lung Cancer
Sevabertinib, a novel reversible HER2-EGFR inhibitor, demonstrated remarkable efficacy in the Phase I/II SOHO-01 study. The objective response rate (ORR) was 64% in pretreated patients and as high as 71% in treatment-naïve patients, with activity also observed in those with brain metastases. With its unique mechanism of action and excellent selectivity, Sevabertinib is poised to reshape the treatment landscape for HER2-mutant non-small cell lung cancer (NSCLC). The Phase III confirmatory study, SOHO-02, has now been officially initiated.

First-Ever! FDA Approves New Drug Paltusotine for Rare Disease
This new non-peptide somatostatin receptor 2 (SST2) agonist has become the first FDA-approved once-daily oral treatment for acromegaly, providing patients with a convenient and effective new treatment option.

Eli Lilly's Inluriyo Approved: A New Treatment for ESR1-Mutated Advanced Breast Cancer
Eli Lilly and Company's oral drug Inluriyo (imlunestrant) has received approval from the U.S. Food and Drug Administration for the treatment of a specific type of advanced or metastatic breast cancer.
